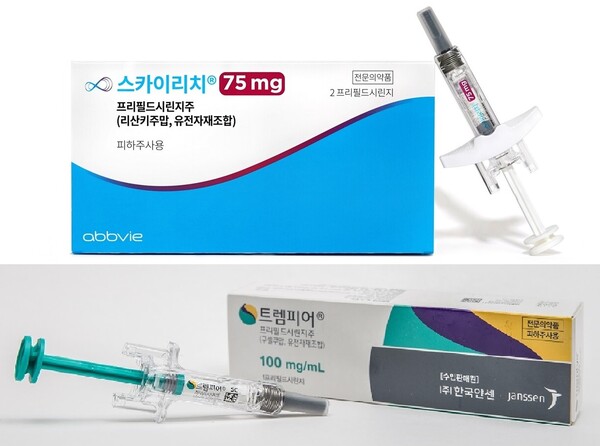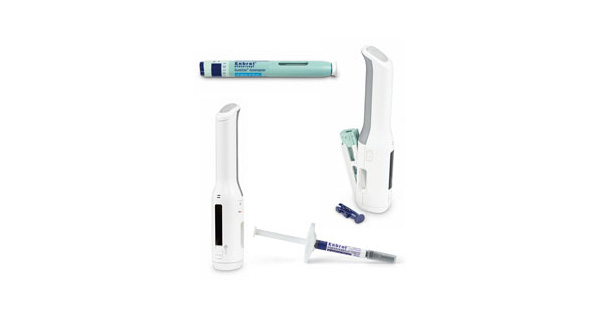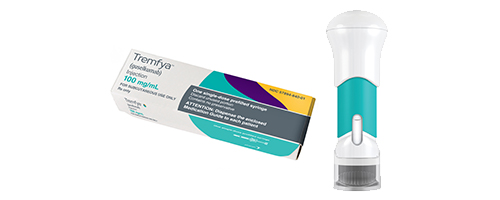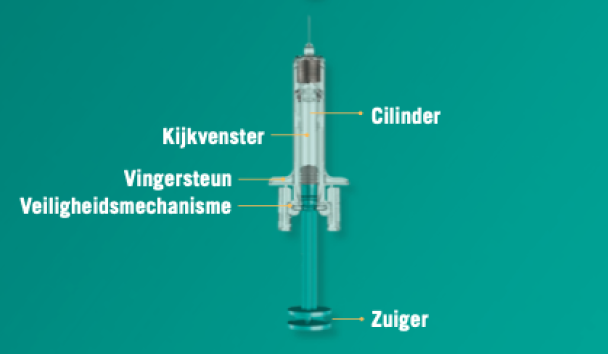
Janssen Announces U.S. FDA Approval of Novel TREMFYA® (guselkumab) One-Press Patient-controlled Injector for Adults with Moderate-to-Severe Plaque Psoriasis
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use TREMFYA safely and effec

Janssen Announces U.S. FDA Approval of Novel TREMFYA® (guselkumab) One-Press Patient-controlled Injector for Adults with Moderate-to-Severe Plaque Psoriasis